The answer is simple: Yes.
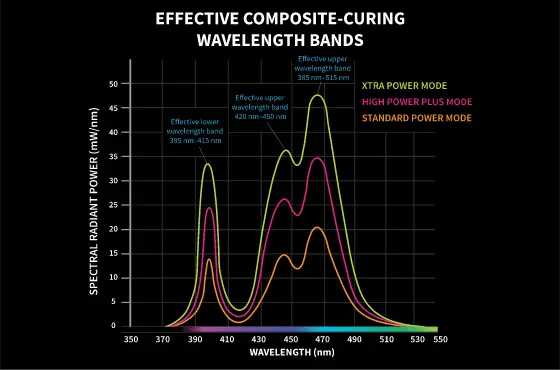
Why? If these are not activated, they will not polymerize. If they do not cure, there is a risk that the restoration will fail. The reason a clinician may think it is okay not to cure all of the material at once is because they have heard that the cure continues after the initial activation. While this is true, as the molecular chains continue to bond 24-72 hours after activation, there is an underlying problem. The photoinitiators must be activated first. If there is a photoinitiator of its own and it does not receive light in the wavelength range 395 – 415, it will NOT activate. Similarly, if camphorquinone does not receive light in the wavelength range 439 – 490, it will not activate.
Many manufacturers of dental products do not disclose the types of photoinitiators they use in their materials. This gives them the flexibility to change them at their convenience, which they do. The truth is that there are many light-curing dental materials that do not use their own photoinitiators or camphorquinone, but use both (co-initiators). This makes the wavelength spectrum of a curing light even more important in today's dental market.
VALO's LED package, with chips in 3 different wavelengths, allows VALO to polymerize all materials containing proprietary photoinitiators and camphorquinone.